
Kevin Miller’s deep brain stimulation surgery helped him overcome almost all the symptoms of Parkinson’s Disease. (Photo: Eric Bronson, U-M Photo Services.)
Kevin Miller spent a lot of time bowhunting this fall. Strapped in his tree stand, he watched flying squirrels play, napped, sat quietly as deer passed within three feet of his tree. He hasn’t shot at anything yet. “I don’t have to,” he says, and you get the sense he’s already found what he was hunting for.Miller, 50, has Parkinson’s disease. For 10 years he coped with the symptoms and side effects—involuntary shaking and jerking, difficulty sleeping, a blank facial expression, difficulty thinking, an unruly and unreliable left arm—as his doctors tried to find a medication and dose to help him manage the disease.When his neurologist told him he was a candidate for Deep Brain Stimulation (DBS)—a set of thin wire electrodes sunk deep into his brain to carry a regulating electrical signal to his misbehaving neurons—Miller didn’t exactly leap at the prospect.
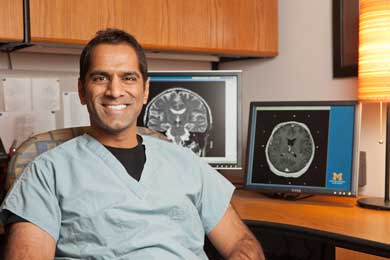
Dr. Parag Patil, who performed the surgery on Kevin Miller, says that DBS is ‘like the oboe that begins a concert,’ coordinating and tuning different parts of the brain. (Photo: Austin Thomason, U-M Photo Services.)
But between the disease and the medications, Miller had progressively given up so much—running, hunting, playing catch with his three kids, the masters degree in medical physics he was working on. Finally, feeling cornered and out of options, he had the surgery on Feb 14, 2011. Miller spent six hours in surgery, awake as U-M neurosurgeon and engineer Parag Patil positioned his DBS probes and neurologist Kelvin Chou painstakingly tested his responses to make sure the signal was reaching the right neurons.”I think of the deep brain stimulator like the oboe that begins a concert,” said Patil. “Before the oboe starts to play everyone’s doing their own thing and it sounds like a lot of noise. But the oboe comes in and soon everyone’s playing together. The neurons are acting in concert and the tremor goes away.”When the unit was connected to its power source (a pacemaker-like device implanted in his chest in a separate surgery), Miller noticed the difference almost immediately.

Daryl Kipke leads a team of U-M alumni in the high-tech startup company NeuroNexus (Clockwise from lower left: Kipke, John Seymour, KC Kong, Hirak Parikh. Photo: Scott Soderberg, U-M Photo Services.)
His movements became smoother; his muscles more coordinated. No longer medicated into a stupor, he continues to rediscover things he once thought were lost forever—the ability to tie his shoes, throw a frisbee, draw a bowstring, sit in his tree stand on a perfect fall day. For patients like Miller, DBS can be life-changing, though it’s not always without side effects.Before the surgery, Miller would wake up at night with his left leg jerking because his medication had worn off. Now he wakes up for no reason and can’t get back to sleep. His voice is softer since the surgery, and unless he concentrates hard on speaking slowly, his thoughts can accelerate into a fast-forward rush of words. “Some people feel it’s not worth it,” Miller said of the surgery, which carries a 1 percent risk of stroke. “But for me the benefits greatly outweigh the side effects or risks. It’s given me back quite a bit of quality of life.”

This probe developed by NeuroNexus is an evolutionary step forward for deep brain stimulation. (Photo: Scott Soderberg, U-M Photo Services.)
Miller’s implants represent the first generation of DBS, the best the field can offer right now. But biomedical engineers are convinced that’s not the best DBS can do. And Michigan—with a long history in brain probe research, wealth of engineering know-how and world-class nanofabrication facilities—is uniquely equipped to drive the technology forward.The university has been at the forefront of neural implant research since the field was little more than a bright idea, starting with the silicon probe developed by now-emeritus engineering professor Ken Wise, who came to U-M in 1974. Biomedical engineering at Michigan has grown up in an uncommonly collaborative atmosphere that continues to generate inventions and fledgling biotech companies. The U-M Office of Tech Transfer has launched 93 startups and licensed more than 800 inventions since 2001. And the university’s Lurie Nanofabrication Facility (LNF), which enjoyed a $65 million renovation in 2008, gives engineers the tools to make things smaller, faster and better. For a fee, small companies can send their technicians to the LNF to use facilities and equipment that they couldn’t hope to afford otherwise.One of those startups, NeuroNexus Technologies, has partnered with the Dutch company Phillips Research to develop what may be the next generation of deep brain stimulation.NeuroNexus, which spun off from the university in 2004, has a scientific team made up of half a dozen Michigan PhDs—neural engineers who’ve either studied under the field’s pioneers or pioneered alongside them. So while the company pays its bills by turning out commercial-grade probes to be used in research labs worldwide, it wasn’t much of a leap to consider the challenge of creating a better, more accurate DBS probe.”For me what’s cool about (working with the brain) is that it just gets better,” says U-M biomedical engineer Daryl Kipke, who’s also president and CEO of NeuroNexus. “As we make improvements in technology, as we do a better job there, we’re able to use it in more sophisticated ways to better interface with the brain. And that leads to more insights and more sophisticated technology.”In a conference room at NeuroNexus’s Ann Arbor offices, Kipke lifts a prototype DBS probe out of its plastic show-and-tell case. It has a thin, jiggly lead about four inches long, made to slide into a patient’s brain and spend decades riding around inside their skull. Like all NeuroNexus probes, it uses thin film technology to pack a lot of high-quality contact points into the device without increasing its size. The prototype in Kipke’s hand—now licensed to a joint venture called Sapiens Steering Brain Stimulation—has more than ten times as may contact points as a conventional DBS.Kipke likens its abilities to a good pair of gloves.”If the goal is to protect your hand and interface with something is a highly tactile way, a well-fitted, well-designed glove could provide that higher-resolution, higher-fidelity experience compared to a mitten,” he said. “More conventional probes are more like mittens.”The current generation of DBS implants have four contact bands that wrap around the electrode like strips of tape around a broom handle. An electrical pulse emits from those bands, creating a uniform “ball” of stimulation. But the brain is a crowded neighborhood, and conventional implants sometimes stimulate cells that don’t need it. That can cause side effects.So instead of bands, the prototype NeuroNexus developed has many smaller contact points, arranged in dots up and down the electrode on all sides. Each can be turned on or off independently, letting doctors “steer” the stimulation field to reach specific cells without bothering others.Sapiens now shoulders the responsibility of getting it to clinical trials and eventually, if things go well, into the hands of neurosurgeons.Patil performs 30-40 DBS surgeries a year, treating not just Parkinson’s Disease, but also obsessive compulsive disorder and two other movement disorders, dystonia and essential tremor. Better technology, he says, could widen the field even more.”Our belief is the more sophisticated lead will allow for better, more accurate stimulation,” said Patil, who has worked with NeuroNexus on this and other DBS technologies. “One thought is we may be able to better control the side effects of stimulation. (And better implants) could lead to DBS being applied to many more diseases.”







James Davis - 1980
I think even more promising than DBS, is a therapy device called MR Guided Focused Ultrasound (MRgFUS). It is a completely non-invaisve technology that uses high intensity ultrasound, under the guidance of MRI to ablate tissue. The company that leads in this area, Insightec, has recently completed a phase 1 trial treating Essential Tremor Disorder. The trial was quite successful. There have been trials for Parkinson’s disease as well.
Reply
Joe Gallagher
I’ve heard that DBS as a treatment modality is mostly effective in decreasing the incidence and severity of tremors, but not so hot in Rx of the other symptoms. Is this a fact or a rumor ?????
Dr. Patil replies: In fact, there are many symptoms that improve. As a rule of thumb, anything that gets better with LevoDopa gets better with DBS. Also the side-effects of Levodopa get better (dyskinesias). Kelvin Chou and I just published a book on DBS for patients and their families, it’s entitled “Deep Brain Stimulation: A New Life for People with Parkinson’s, Dystonia, and Essential Tremor.”
Reply
Jason Homes
I think this is the right approach and way of thinking. One should never give up in front of the diagnose and keep on having an active life, as he did. I really admire people with strong will and spirit.
Reply