Alzheimer’s dementia
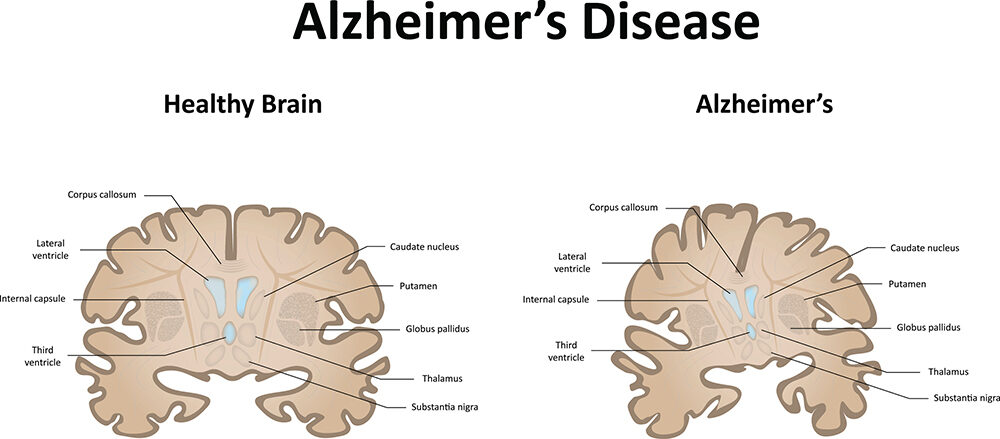
Dementia is an overall term for a particular group of symptoms that include difficulties with memory, language, problem-solving, and other thinking skills that affect a person’s ability to communicate and perform everyday activities. Alzheimer’s disease is one of many causes for dementia that include excessive accumulation of the protein fragment beta-amyloid and an abnormal form of the protein tau, as well as damage to and destruction of brain neurons. The brain changes in Alzheimer’s disease are the most common contributor to dementia – hence the name Alzheimer’s dementia.
The word ‘dementia’ first appeared around 600 A.D. Saint Isidore (560–636 A.D.), the archbishop of Seville, appears to have been the first to use the term in his book, Etymologies. The word originates from Latin, and is formed from the prefix ‘de,’ which means a deprivation or loss; root ‘ment,’ which means mind; and suffix ‘ia,’ which indicates a state. Thus, dementia refers to ‘a state out of mind.’
Explaining how and why specific brain-neuronal changes occur in individuals with dementia has been elusive. The most common explanation has been age and genetics. However, new research now suggests that other factors, including certain lifestyle habits, also may contribute to brain changes leading to dementia. These findings have sparked new efforts to find possible dementia treatment options previously not appreciated. This is exciting news!
Dementia prevalence
Alzheimer’s begins 20 years or more before symptoms develop. Thus, prevalence rates are underreported. Some studies indicate that underdiagnosis is higher among Black and Hispanic older adults.
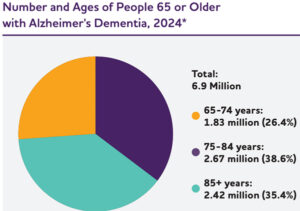
An estimated 6.9 million Americans aged 65 and older are currently living with Alzheimer’s dementia. Seventy-three percent are age 75 or older. Of the total U.S. population, about 1 in 9 people age 65 and older has Alzheimer’s dementia, and the prevalence increases with age. While individuals younger than 65 can also develop Alzheimer’s dementia, studies of younger-onset dementia are limited. Nevertheless, researchers predict about 110 of every 100,000 people aged 30-64 years, or about 200,000 Americans in total, have younger-onset dementia.
Dementia symptoms
Alzheimer’s dementia symptoms vary from person to person. In general, memory problems are the first sign and often include a decline in non-memory aspects of cognition – finding the right word, trouble understanding visual images and spatial relationships, and impaired reasoning or judgment. As the disease progresses, symptoms become more severe, including increased confusion and behavior changes.
Preclinical Alzheimer’s dementia
The first signs of pre-clinical Alzheimer’s dementia usually include complex brain changes that include formation of brain amyloid plaques or tau tangles and can be detected a decade or more before memory and thinking problems appear.
Mild Alzheimer’s dementia
A person with mild Alzheimer’s dementia may seem healthy but has more and more trouble making sense of the world around them. The realization that something is wrong often comes gradually to the person and their family.
— Symptoms: Memory loss that disrupts daily life. Poor judgment leading to bad decisions; loss of spontaneity and sense of initiative; losing track of dates or knowing current location; taking longer to complete normal daily tasks; repeating questions or forgetting recently learned information; trouble handling money and paying bills; challenges in planning or solving problems; getting lost; losing things or misplacing them in odd places; difficulty completing tasks such as bathing; mood and personality changes; increased anxiety and/or aggression.
Moderate Alzheimer’s dementia
Intensive supervision and care may become necessary at this stage. Alzheimer’s is often diagnosed at this stage.
— Symptoms: Increased confusion and memory loss, such as forgetting events or personal history; withdrawal from social activities; inability to learn new things; difficulty with language and problems with reading, writing, and working with numbers; difficulty organizing thoughts and thinking logically; shortened attention span; problems coping with new situations; sleeping more during the day and being restless at night; difficulty carrying out familiar, multistep tasks, such as getting dressed; occasional problems recognizing family and friends; hallucinations, delusions, and paranoia; impulsive behavior, such as undressing at inappropriate times or places, or using vulgar language; inappropriate emotional outbursts; restlessness, anxiety, tearfulness, wandering about in the afternoon or evening; repetitive statements or movement; occasional muscle twitches.
Severe Alzheimer’s dementia
People with severe Alzheimer’s dementia cannot communicate and are entirely dependent on others for their care. The person may be in bed most or all of the time as their body shuts down.
— Symptoms: Inability to communicate; no awareness of recent experiences or surroundings; weight loss with little interest in eating; seizures; general physical decline; difficulty swallowing; groaning, moaning, or grunting; increased sleeping; loss of bowel and bladder control.
Alzheimer’s dementia non-modifiable risk factors
The vast majority of people who develop Alzheimer’s dementia are age 65 or older. This is called late-onset Alzheimer’s dementia. Experts believe that Alzheimer’s dementia, like other common chronic diseases and conditions, develops as a result of multiple factors rather than a single cause. Exceptions are cases of Alzheimer’s related to Down syndrome, and rare cases related to specific genetic mutations.
Major non-modifiable risk factors
 Age: Age represents the most significant risk factor for the disease. The percentage of people with Alzheimer’s dementia increases dramatically with age, but age alone is not sufficient to cause Alzheimer’s dementia.
Age: Age represents the most significant risk factor for the disease. The percentage of people with Alzheimer’s dementia increases dramatically with age, but age alone is not sufficient to cause Alzheimer’s dementia.- Genes: In 2022, researchers identified 31 new genes directly related to Alzheimer’s dementia. Of these, the APOE4 has the strongest impact association. APOE4 provides the blueprint for the protein cholesterol transporter in the blood. In addition, those with the APOE4 gene are more likely to have beta-amyloid accumulation and Alzheimer’s dementia at a younger age. Thus, the APOE4 gene is widely recognized as an Alzheimer’s genetic risk factor and is implicated in 60-80% of all Alzheimer’s dementia cases.
- Family History: Individuals who have or had first-degree relatives with Alzheimer’s dementia are more likely to develop the disease than those who do not have Alzheimer’s dementia in the family. Those who have more than one first-degree relative with Alzheimer’s dementia are at even higher risk. When diseases run in families, genetics and shared non-genetic factors (for example, access to healthy foods and habits related to physical activity) may play a role.
Although age, genetics, and family history cannot be changed, we can modify some risk factors to reduce the risk of cognitive decline and dementia. Examples of modifiable risk factors are physical activity, smoking, education, staying socially and mentally active, blood pressure, and diet. Research suggests that up to 40% of dementia cases may be attributable to these modifiable risk factors. Notable, a 2022 study found that nearly 37% of cases of dementia in the U.S. were associated with eight modifiable risk factors, the most common being midlife obesity, followed by physical inactivity.
Alzheimer’s dementia modifiable risk factors
- Cardiovascular Health Factors: Brain health is affected by heart and blood vessel health. Although the brain makes up just 2% of body weight, it consumes 20% of the body’s oxygen and energy supplies. A healthy heart ensures enough nutrient-rich blood reaches the brain. Other factors that increase the risk of cardiovascular disease also associate with a higher dementia risk. These include hypertension, diabetes, and smoking.
- Sleep: Researchers have found that an important function of sleep is the removal of toxic beta-amyloid and other substances from the brain. Inadequate or poor sleep has been shown to compromise the brain’s ability to remove toxins, enabling levels of toxins to remain elevated. Some evidence suggests the relationship between sleep and Alzheimer’s disease is bidirectional: Poor sleep increases one’s risk of Alzheimer’s, and Alzheimer’s brain changes may increase the risk of inadequate sleep.
- Air Pollution: There is rapidly emerging evidence on how exposure to environmental toxicants, especially air pollution, relates to increased dementia risk. Fine particulate matter (PM) air pollution — tiny solid particles and liquid droplets generated by fuel combustion, fires, and processes that produce dust — consistently have been shown to have the most significant dementia-risk impact.
Putting exercise and plant-based eating to the test
It is well known that specific lifestyle changes, including a plant-based diet and regular exercise, can reverse the progression of heart disease, the number-one killer worldwide. Changes in diet and exercise can open the arteries without drugs and/or surgery. And the same plant-based diet and exercise program has been shown to reverse the course of early-stage prostate cancer. Evidence shows these changes also elongate our telomeres (structures made from DNA sequences and proteins found at the ends of chromosomes), suggesting an anti-aging effect as well.
Researchers who tested and showed the power of plant-based eating and regular, moderate exercise are seeking to demonstrate how the same interventions can delay the onset, reduce the symptoms, and even reverse Alzheimer’s dementia for many individuals.
This is a big deal!
Related research has demonstrated that high circulating blood cholesterol is unanimously recognized as a major risk factor for the development of Alzheimer’s dementia. Those individuals with a blood cholesterol of 225 or more have 25% greater odds of having amyloid plaques in the brain 10-15 years later, and higher levels of the APOE4 gene, which codes for the major cholesterol carrier inside the brain. But having high APOE4 gene levels does not necessarily confer Alzheimer’s disease. There is impressive research showing those with high APOE4 gene rates, but who have low levels of cholesterol due to genetic variation (or by following a plant-based diet), have some of the lowest rates of Alzheimer’s dementia. Perhaps this is the reason that vegetarians may be three times more likely not to have dementia later in life.
Now, researchers have published results of an important randomized-control, phase-2 clinical trial to determine if the progression of Alzheimer’s dementia may be slowed, stopped, or even reversed by a comprehensive, multimodal, intensive lifestyle intervention after just 20 weeks, compared to a usual-care randomized control group.
Scientists randomized 50 men and women with verified early-stage Alzheimer’s into one of two groups for a 20-week intervention program. The first group made no lifestyle changes, while the second group ate a whole-food, plant-based diet (with supplements like vitamin B12), exercised moderately (walking 30 minutes per day), practiced stress management (relaxing, breathing exercises), and got group support (over Zoom).
The researchers performed standard tests for cognition and function before and after the 20-week intervention. They also measured objective experimental biomarkers of disease progression.The results are noteworthy. On the Clinical Dementia Rating Global scale, which is used to stage the severity of dementia, the control group (who did not make any changes in diet and exercise) continued to decline. None showed any improvement. In contrast, about 40% of those in the diet and lifestyle group appeared to be getting better within five months of eating and living more healthfully. Most noteworthy, the more the participants in the diet and lifestyle group complied with the recommendations, the more they experienced significant, beneficial impacts on their cognition and function.
As with every study, some limitations need to be highlighted. In drug trials, some participants receive a placebo for comparison with the drug’s effects. In studies regarding diet and lifestyle changes, however, researchers have to rely on subjective questions like, “How has your memory been?”
That said, the Alzheimer’s dementia researchers also measured objective biomarkers of disease progression. They observed a positive trajectory in the intervention group and a worsening in the control group, with the same apparent dose-response effect (the more the subjects improved their diet and lifestyle, the more dramatic the effect).
When comparing these findings with standard Alzheimer’s dementia treatment options, the results clearly show that lifestyle changes are superior. Moreover, drug treatment options can cost upwards of $26,500 annually and also could cost U.S. taxpayers an additional $82,500 per patient annually for genetic tests and frequent brain scans, safety monitoring, and other care options covered by Medicare and other public assistance options.
The bottom line
For the first time, evidence shows that intensive lifestyle intervention, including plant-based eating and consistent moderate exercise, is effective in preventing, arresting, and reversing the progression of Alzheimer’s dementia.
This is remarkable.
I sincerely hope you join me in eating more plants and engaging in moderate exercise every day. This is how you health yourself!
References
Albert, M.S., et al. “The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease.” Alzheimer’s Dementia. 2011;7:270.
Alzheimer’s disease facts and figures: 2024
Barnard, N.D., et al. “Dietary and lifestyle guidelines for the prevention of Alzheimer’s disease.” Neurobioliogy of Aging. 2014;35 Suppl 2:S74-S78.
Cutuli, D., et al. “Physical exercise as disease-modifying alternative against Alzheimer’s disease: A gut–muscle–brain partnership.” International Journal of Molecular Sciences. 2023; 24(19):14686.
de la Torre, J.C. “Alzheimer’s disease is incurable but preventable.” Journal of Alzheimer’s Disease. 2010;20(3):861.
Dhana, K., et al. “Healthy lifestyle and cognition in older adults with common neuropathologies of dementia.” JAMA Neurology. 2024.
Dhana, K., et al. “Healthy lifestyle and the risk of Alzheimer dementia: Findings from 2 longitudinal studies.” Neurology. 2020;95(4):e374–83.
Ding, H., et al. “Plants, plants, and more plants: Plant-derived nutrients and their protective roles in cognitive function, Alzheimer’s disease, and other dementias.” Medicina (Kaunas). 2022;58(8):1025.
Hendrie, H.C., et al. “Prevalence of Alzheimer’s disease and dementia in two communities: Nigerian Africans and African Americans.” American Journal of Psychiatry. 1995;152(10):1485.
Hu, X., et al. “Association between abnormal lipid metabolism and Alzheimer’s disease: New research has revealed significant findings on the APOE4 genotype in microglia.” Bioscience Trends. 2024 Jun 6;18(2):195.
Livingston, G., et al. “Dementia prevention, intervention, and care: 2020 report of the Lancet Commission.” Lancet. 2020;396(10248):413.
Morris, M.C., et al. “Associations of vegetable and fruit consumption with age-related cognitive change.” Neurology. 2006;67(8):1370.
Ngandu, T., et al. “A 2-year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomized controlled trial.” Lancet. 2015;385(9984):2255.
Ornish, D., et al. “Can lifestyle changes reverse coronary heart disease? The Lifestyle Heart Trial.” Lancet. 1990;336(8708):129.
Ornish, D., et al. “Effect of comprehensive lifestyle changes on telomerase activity and telomere length in men with biopsy-proven low risk prostate cancer: 5-year follow-up of a descriptive pilot study.” The Lancet Oncology. 2013;14(11):1112–20.
Ornish, D., et al. “Effects of intensive lifestyle changes on the progression of mild cognitive impairment or early dementia due to Alzheimer’s disease: A randomized, controlled clinical trial.” Alzheimer’s Research & Therapy. 2024;16(1):122.
Ornish, D., et al. “Intensive lifestyle changes for reversal of coronary heart disease.” JAMA. 1998;280(23):2001–7.
Ornish, D., Weidner, G., Fair, W.R., et al. “Intensive lifestyle changes may affect the progression of prostate cancer.” Journal of Urology. 2005;174(3):1065.
Rajan, K.B., et al. “Population estimate of people with clinical A.D. and mild cognitive impairment in the United States (2020-2060).” Alzheimers Dementia. 2021;17(12):1966.)
Rosenberg, A., et al. “Multidomain lifestyle intervention benefits a large elderly population at risk for cognitive decline and dementia regardless of baseline characteristics: The FINGER trial.” Alzheimers Dementia. 2018;14(3):263–70.
Yang, H.D., et al. “History of Alzheimer’s disease.” Dementia and Neurocognitive Disorders. 2016 Dec;15(4):115.
Yu, J.T., et al. “Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomized controlled trials.” Journal of Neurology, Neurosurgery, and Psychiatry. 2020;91(11):1201–9.
Lead image: iStock.



Catherine Church - 1967
I have taken classes with Dr. Barnard and Dr. Ornish as well as many others who offered week-long classes, once a year on board a ship. It was called Holistic Holiday at sea. We were 50% of the travelers, and we were there to enjoy to all sorts of events regarding all of the lifestyle things we should know about to enhance our health. Even the meals were separated including our own time in the restaurant that served only vegan foods. What a marvelous event. It has greatly improved my life style and we went back 9 times–once each summer. I am hopeful that this is why I was able to slow down my chance of Alzheimers. I still have to take a daily set of pills to further slow my cognitive decline. I am now 79 and still feeling very well, although I do still have mild cognitive decline.
Reply
Catherine Hoffman Church - 1967
Yes, I do have the APOE4 gene. But I did not realize until I was in my late 60’s that I could slow that down if I modified my life style. I was delighted when we started with the week-long the classes with John McDougall in California. That got us started on the right way. Then I had a pet scan and there they were — amyloid plaques in my brain! THEN, we were interested in the things that others were doing so we took classes aboard a ship where a special event was offered about being healthier, called Holistic Holiday at Sea. It included a lot of things about food with Dr. Barnard and Dr. Ornish and many other professionals and speakers that applied to being healthy. My husband and I attended seven of these weeks and it has been a life saving set of events. I am still doing well but I now take appropriate supplements and a medication (Memantine and Donepezil). I am now 79 and still functioning fairly well so far with only mild cognitive decline. Thank you Dr. Barnard et. al.
Reply
Russell Lyons - 1983
I presume the study discussed in the last section is this one:
Ornish, D., et al. “Effects of intensive lifestyle changes on the progression of mild cognitive impairment or early dementia due to Alzheimer’s disease: A randomized, controlled clinical trial.” Alzheimer’s Research & Therapy. 2024;16(1):122.
There are a couple of additional limitations of this study that I think are worth acknowledging.
First, 8 supplements were used in the treatment group, some with several “effective” ingredients. These were provided because the investigators believe there is evidence of their positive effect on AD. Since several interventions at once were part of the treatment, one cannot know which, if any, were responsible for the results.
Second, the intervention and testing lasted only 20 weeks. Thus, the study gives no evidence of long-term difference. For example, it could be that the exercise regime increased physical health, which in turn led to increased blood flow to the brain and thus improved scores, an effect that might not slow the progression of AD.
I don’t mean to discourage a vegan diet nor exercise, both of which are important to me personally. However, I think it is important to be clear about what is known.
Reply