While the terms “biotechnology” and “genetic modification” or “genetic engineering” often are used interchangeably, they represent quite different processes and outcomes.
Biotechnology is the general term that refers to using organisms or their components, such as enzymes, to make products that include wine, cheese, beer, and yogurt. Genetic modification (GM), on the other hand, represents a special set of technologies that alter the genetic makeup of animals, plants, or bacteria that would not take place naturally, bringing about alterations in genetic makeup and properties. Combining genes from different organisms is known as recombinant DNA technology, and the resulting organism is said to be “genetically modified,” “genetically engineered,” or “transgenic.” GM products include medicines and vaccines, foods and food ingredients, feeds, and fibers.
Most people welcome GM products in the health and medicine field where genetically altered materials produce life-saving drugs. Not so when it involves our food.
Consuming GM foods constitutes the largest uncontrolled human nutrition experiment ever performed.
Introduction of GM foods into the world’s food supply at unprecedented levels constitutes one of the largest human nutrition experiments ever performed. Foods are being distributed that have not been adequately tested for long-term human safety or consequences, without a control group, without approval by those participating in the experiment (it’s virtually impossible to know if you are eating GM foods since labeling is not required in the U.S.), and without adequate oversight or a way to stop the experiment if something were to be proven wrong (as is the case in all human experiments).
Genetic engineering: Promises and findings
Since introduction of GM foods, proponents argue they have the potential to enhance taste and quality; reduce maturation time and hence time-to-market; increase nutrients, yields, and stress tolerance; improve resistance to disease, pests, and herbicides; and produce “new” products and growing techniques. While many of these promises have been achieved, not everything is so rosy.
A growing body of evidence connects GMs with various health problems, environmental damage, and violation of farmers’ and consumers’ rights.
In nearly 50 countries around the world, including Australia, Japan, and all countries in the European Union, there are significant restrictions or outright bans on the production and sale of GM food.
The process of inserting a genetically modified gene into the DNA of a plant cell is crude, uncontrolled, and imprecise, and causes mutations in the plant’s DNA blueprint. These mutations can alter functioning of the natural genes of the plant in unpredictable and potentially harmful ways. The American Academy of Environmental Medicine 2009, after reviewing numerous studies on GM foods, recommended that physicians advise patients to avoid them when possible (www.aaemonline.org/gmopost.html).
Before the Food & Drug Administration (FDA) allowed GMs into food without labeling, FDA scientists had repeatedly warned that GM foods could create unpredictable, hard-to-detect side effects, including allergies, toxins, new diseases, and nutritional problems. They urged long-term safety studies, but were ignored.
Since then, different research findings on GM foods have revealed the following:
- Thousands of sheep, buffalo, and goats in India died after grazing on GM cotton plants.
- Mice eating GM corn for the long term had fewer, and smaller, babies.
- More than half the babies of mother rats fed GM soy died within three weeks, and were smaller.
- Testicle cells of mice and rats on a GM soy diet changed significantly.
- By the third generation, most GM soy-fed hamsters lost the ability to have babies.
- Rodents fed GM corn and soy showed immune system responses and signs of toxicity.
- Soy allergies skyrocketed by 50 percent in the U.K. soon after GM soy was introduced.
Most importantly, unintended, uncontrolled mutations occur during the GM process, resulting in unexpected changes in the crop and foods produced from it. These unexpected changes can create more opportunities to interact with other organisms and the environment, generating even more unintended and unpredictable side effects.
How to avoid GM foods

- Buy Organic. Organic producers cannot intentionally use GMOs.
- Look for “Non-GMO Project Verified” seals.
- Avoid at-risk ingredients. If it’s not labeled organic, or doesn’t have a Non-GMO Project Verified seal, then avoid food products made with the following crops since they are mostly genetically modified in the U.S.: • Corn (corn flour, meal, oil, starch, gluten, and syrups; sweeteners such as fructose, dextrose, and glucose; and modified food starch)• Soy (soy flour, lecithin, protein, isolate, and isoflavone)• Canola (canola oil, also called rapeseed oil)• Cottonseed oil• Sugar (avoid anything not listed as 100 percent cane sugar)• Aspartame (the artificial sweetener also known as NutraSweet and Equal, which is derived from GM microorganisms)
- Avoid hidden GM products, including baking powder, caramel color, lecithin, malt sugar, soy milk, soy sauce, tempeh, whey, whey powder, and sorbitol.
Coming soon to your local fish market?
The next chapter in the unfolding drama of genetically engineered food is the private development of genetically engineered fish raised under industrial aquaculture conditions. The FDA, entrusted with oversight of GM animals, is currently considering whether to approve a GM salmon (referred to as the “AquAdvantage” salmon) engineered to grow faster and produce more and better meat.
The transgenic AquAdvantage salmon is an Atlantic salmon that has had its DNA spliced with a growth hormone gene from the unrelated Chinook salmon and anti-freeze DNA from an Arctic eelpout that stimulates year-round growth, thus creating a fish that its developer claims can grow up to twice as fast as conventionally raised Atlantic salmon. If approved, the AquAdvantage salmon would be the first-ever GM animal approved for human consumption.
GM salmon food safety concerns
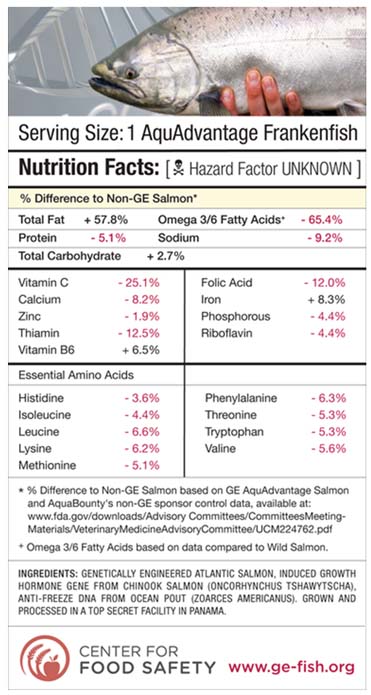 The FDA has yet to require or conduct any studies to assess possible long-term health impacts of eating GM animals; however, several independent studies have produced findings that leave cause for serious concern. For instance, the routine use of antibiotics to cont
The FDA has yet to require or conduct any studies to assess possible long-term health impacts of eating GM animals; however, several independent studies have produced findings that leave cause for serious concern. For instance, the routine use of antibiotics to cont
rol disease in factory farm operations (like the AquAdvantage salmon conditions) may adversely impact human health. Research suggests transgenic fish may be even more susceptible to diseases than conventionally farmed fish.
Increased disease susceptibility could mean transgenic fish may then require more antibiotics. Human health also could be jeopardized as a result of antibiotic-resistant bacteria and exposure to certain classes of antibiotics that may cause allergic reactions.
Genetically engineered salmon lack many of the beneficial qualities that non-GM salmon boast, specifically, wild salmon have 189 percent higher levels of beneficial omega fats than GM salmon.
In another study, concerns were expressed that both toxins and growth hormones had a high potential to end up in consumers’ bodies. Also, it has been shown that genetically engineered salmon possess 40 percent higher levels of the hormone called IGF-1 (insulin-like growth factor 1), which has been shown to increase the risk of certain cancers.
The graphic below shows an example of comparisons between the AquAdvantage “Frankenfish” and non-GM salmon, based on the best available evidence to date.
Do we really need such fish?
References:
• Antoniou M., et al. (2012) “GMO Myths and Truths: An Evidence-based Examination of the Claims Made for the Safety and Efficacy of Genetically Modified Crops.” Earth Open Source.
• Centre for Aquaculture and Environmental Research (2009) “Ecological Risk Assessment of Transgenic Salmon.” Study commissioned by the European Union, the Swedish Research Council Fromas, and the University of Gothenburg. Vancouver, Canada.
• Genetically Engineered Salmon: The Next Generation of Industrial Aquaculture. Food Safety Review: A Publication of the Center For Food Safety
• Ian Phillips, et al. (2003) “Does the Use of Antibiotics in Food Animals Pose a Risk to Human Health? A Critical Review of Published Data.” Journal of Antimicrobial Chemotherapy. vol. 53, 28–52.DOI: 10.1093/jac/dkg483.
• Moschos, S. (2002) “The Role of the IGF System in Cancer: From Basic to Clinical Studies and Clinical Applications.” Oncology, vol. 63(4).
• Non GMO Project• U.S. Food and Drug Administration Center for Veterinary Medicine, Veterinary Medicine Advisory Committee. “Briefing Packet: AquAdvantage Salmon.” Sept. 20, 2010.
• U.S. Food and Drug Administration Center for Veterinary Medicine. “Draft Environmental Assessment: AquAdvantage Salmon.” May 4, 2012 (published Dec. 26, 2012), p. 86.
• Yu H. and T. Rohan. (2000) “Role of the Insulin-Like Growth Factor Family in Cancer Development and Progression.” Journal of the National Cancer Institute, vol. 92(18).



Ramona Bashshur - 1984
Excellent article. It is also worth noting that current FDA policy which finds GM foods to be the same as their non-GM counterparts, is 2 decades old and should be revisited. It was adopted in furtherance of goals other than food safety; namely, promotion of the biotech food industry.
Reply
Rork Kuick - 1983
Very one-sided and many criticism are possible.
1) Every new variety is a nutrition experiment, GMO or not. And I think there are ways to stop a GMO if found harmful – that wasn’t explained.
2) Breeding plants in ordinary ways also is chaotic. Irradiating strains to obtain many random new mutations is done, but not considered GM.
3) Follows by cherry-picked studies. I can do that too. I don’t have time to knock down every one I think doubtful.
4) Sometimes compares GM salmon not to other farm-raised salmon, but rather wild-caught fish. No fair.
5) American Academy of Environmental Medicine sounds cool, eh? Not recognized by the American Board of Medical Specialties though. Listed as a questionable organization and dubious certifying board on Quackwatch.
6) Two references to IGF-1 in cancer, but is there anything about eating IGF-1 in food in either article? Digestion alters proteins ya know.
I’m not saying everything’s great with GMOs, but this article is taking liberties, and lacks balance.
Reply
Kari Dumbeck
I have Gluten and about 30 food allergies. I love this article and thank you for it. I have spent more time and money with the holistic medical fields because main stream medicine I have found really does not want to heal people but push drugs. The current practice of treating symptoms rather than the causes is deplorable. For years now I have read over and over again how GM foods are destroying us and that many countries will not buy our food. Considering how sick we have become it’s not surprising. It’s all over the news that we are the sickest nation with the biggest health budget but it’s gotten us no where. I will be using your article as an example at my next staff meeting which will be about making healthier choices. Thank you and keep up the good reporting.
Reply
David Krause - 1962 BS, 1986 PhD
We read “. . . different research findings on GM foods have revealed the following:” Seven examples are given, but not a single reference or citation that would allow the reader to personally check the source. Why not?
Reply
David Miller - MBA 1982
One aspect of GM that does not seem to be discussed is the process in living organisms of extracting protein molecules from food and incorporating them into protein sequences (read: RNA and DNA) in the living organism. Protein processing mechanisms on a sub-cellular level may “see” protein chains that look like what they need for their current “project.” If a few proteins in a chain–whether associated with the GM organism’s shape, resistance to drout, or size–are differently arranged, the rearranged sequence may be reproduced in subsequent generations of the chain, and hence in the cell. Since we cannot predict where within the ingesting organism these altered sequences will appear, we cannot predict their effects. Without extensive, multigenerational testing and observation of the ingesting organisms, on macro and micro levels, no one can really know how any GM sequence affects ingesting organisms and their offspring.
Perhaps we as a society are so committed to an evolutionary paradigm that we genuinely welcome the expectation of premature death–our own and that of our children–in the hope that a few of our species will adapt and survive while most of us will sicken and die. In that case, let’s have even more experimentation with genetic engineering. Otherwise, as individuals and as a civilization, we need to reject genetically modified products as food for any organism.
Reply
Margaret Anderson - 1970
I am surprised that the US is so far behind other countries on this issue. Thank you for a clear explanation of what I need to look for in the store.
Reply
Gregory Palen - 1975 BBA
I am a farm owner and have made a life career in animal genetics, first in marketing and now in gene selection consulting.
I am sympathetic to the views in this article, as I believe we are seeing negative effects on food animals from use of GMO derived feed crops, especially in ruminant species.
What the layman technologist needs to grasp is that everything that biochemical companies have done via the recombinant processes is also able to be done by patient, persistent genetic selection within the specie– and thus the issues arising from specie-foreign gene insertion are avoided.
All the arguments in favor of these expensive recombinant technologies are based on flawed Malthusian estimates of future food shortage. In fact, existing conventional and organic food production utilizing the available arable land is more than capable of feeding the projected world population growth. On an economic level, organic production methods are more relevant to feeding of third world societies, as it allows them to recycle their local nutrients while utilizing yield gains made from the traditional genetic selection for crop seeds and food producing animals, both of which remain available at lower net cost than the more specialized GMO varieties.
Reply
Mark Andrews - 1984
“A growing body of evidence connects GMs with various health problems, environmental damage, and violation of farmers’ and consumers’ rights.”
This is a totally unsubstantiated claim, easily refuted by the many, many studies by truly reputable scientific and regulatory bodies. It’s disappointing to see this sort of decidedly unscholarly regurgitation associated with my supposedly prestigious Alma mater.
For examples of more reasoned thoughts on the subject, please refer to the following:
http://reason.com/blog/2013/02/22/ronald-bailey-parses-the-top-five-lies-a
http://www.marklynas.org/2013/01/lecture-to-oxford-farming-conference-3-january-2013/
Reply
Susan deGroot - 1973
I am now a mental health therapist in Marquette. Michigan and am passionate about at least getting genetically modified food labeled if not out of our food chain. After extensive personal reading, I believe that many of the societal health and mental health issues we are seeing (eg. autism, ADD, ADHD, etc.) are increasing out to previously unheard of levels are because of what we have done to our food in this country. Ignorance is not bliss. It is a personal tragedy waiting to happen. I applaud all efforts to educate anyone and everyone on this subject.
Reply
Ramona Raybin
Unfortunately for us all, the FDA is under the thumb of Big Agribusiness. The small farmer and organic farmers voices are not heard at the FDA and they have been screaming “foul” for quite some time. Until we demand accountability for the safety and efficacy of our food supply, we can expect increasing adverse affects and I suspect some food “disasters” from GM foods but cynic that I am, Big Agribusiness will do everything in their power to ensure the FDA suppresses the facts.
Reply
J Christ - 9
It should be banned all together. It’s ridiculous to even do it. There’s nothing wrong with an orange. The cucumber is perfect as it is. Still we wanna change them? Why?
No take a moment to really think about what Gene Manipulating really is.
Would you genetically manipulate your unborn child? That would then escalte to all your offspring from then on, being genetically manipulated. And you wouldn’t know the consecuenses.
Why would you do it to nature then?
Best regards,
J. Christ
P.s. We can’t even figure out why grass grows – yet still we want to manipulate it?
Reply
Susana Curatolo - 20
Perhaps that is why the NOP really does not want to talk about it?
Reply